|
It is an older term used to describe the element and has its origins in the word "alkali." Like other elements in the first column, potassium is a member of the alkali group with sodium and cesium. The cationic form of potassium combines with different anions to form salts. Like other alkali metals, it has a single valence electron that is easily removed, creating a cation. 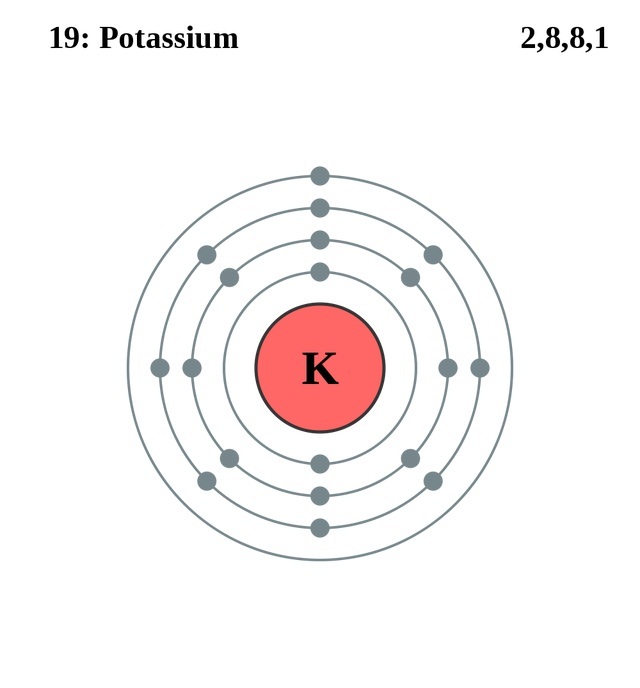
The electron configuration of potassium is Ar 4s 1. It is an alkali metal with atomic number 19. We know where the English name potassium came from, but what about that "K"? The K represents the word Kalium. In the periodic table, potassium (K) is in Group 1 and Period 4. Like sodium (Na) and gold (Au), the symbol is not an abbreviated form of the element's English name. Potassium is a little tricky to remember on the periodic table because the symbol is "K". Once isolated, Davy found potassium to be one of the softer metals with a silver color. It is the most abundant intracellular cation. It is a mineral that your body needs to work properly, and it is a type of electrolyte. It’s electron configuration is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 1. 
In fact, potassium is never found by itself in nature. Potassium is a chemical element with the symbol K. Even though we have been using potash for centuries, Davy officially discovered potassium in 1807. That compound that has been used for hundreds of years and has the chemical formula KOH. Potassium chloride - is also known as potassium salt and is used to produce medication to treat low blood potassium levels. The name potassium comes from a substance called Potash. The first element of the fourth row (period) is potassium.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
